|
6/8/2023 0 Comments Ammonium ion bonding
The strength of cation–anion affinity depends on the distance, the polarity of the solvent and the ionic strength. A typical example of a salt bridge is the ammonium ion carboxylate ion pair. In salt bridges, additional hydrogen bonds are formed. Ĭoulombic interaction attracts cations and anions. A relevant example is the binding of acetylcholine ( 3) in biological systems. The cation–π-bond is an important motif for the recognition of quaternary ammonium ions. The binding energies are between 42 and 92 kJ/mol in the gas phase. Ammonium–π-interactions were experimentally investigated in detail as well as by ab initio calculations and are mainly based on electrostatic interactions. who showed that binding of potassium ions to benzene and water in the gas phase is of similar energy. The first experimental evidence of interactions between cations and aromatic π-systems came from Kebarle et al. Quaternary ammonium ions cannot be bound by hydrogen bonds. Gas phase energies range from 22 kJ/mol (neutral hydrogen bonds between water molecules) up to 163 kJ/mol (anionic F–H–F − complex). If exposed to a competing solvent, a single hydrogen bond cannot contribute much binding energy. Crystal structures mainly show a linear arrangement of the three atoms but bifurcated hydrogen bonds can also be observed. Hydrogen bonds are formed from the strongly polarized N +–H bonds to a free electron pair of an electronegative atom (O, N, F). 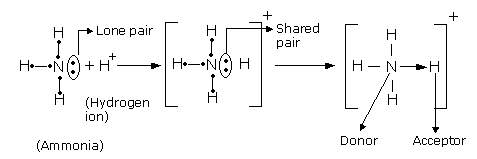
For the recognition of ammonium ions, three types of interactions, mostly acting simultaneously, are typically the most important: Selective ligand-protein receptor binding relies typically on a number of specific interactions between two or more molecules. Therefore the investigation of ammonium ion recognition is of considerable fundamental and practical interest. The understanding of alkylammonium recognition in the dopamine ( 2) class of neurotransmitters is key to the development of tools to study these systems. Malfunction of dopamine-responsive neurons has been implicated in a number of disease conditions including Parkinson’s disease. Once it has bound to these particular peptides, they are no longer available for construction of the bacteria’s cell wall causing their cell death. The inhibition of biological processes is also addressed by molecular recognition involving amino acids and peptides: The antibiotic vancomycin binds selectively with its terminal lysyl- R-alanyl- R-alanine residues in bacterial cells through several hydrogen bonds. An example is the binding of histamine ( 1) to the human H 1 receptor, which results in lower blood pressure and dilatation of blood vessels or plays a primary role for allergic response. As in all biological regulatory processes, selectivity of recognition is of key importance for subsequent steps and cellular response. The interaction of small ammonium ion bearing compounds with protein receptors is important for biological signal transduction processes. 
In our conclusion we compare the strengths and weaknesses of the different types of ammonium ion receptors which may help to select the best approach for specific applications. The molecular recognition of ammonium ions in amino acids is included and the enantioselective binding of chiral ammonium ions by synthetic receptors is also covered. In this review we discuss the different classes of synthetic receptors for organic ammonium ion recognition and illustrate the scope and limitations of each class with selected examples from the recent literature. Typical intermolecular interactions are hydrogen bonds, electrostatic and cation–π interactions, hydrophobic interactions or reversible covalent bond formation. The type of host compounds for organic ammonium ion binding span a wide range from crown ethers to calixarenes to metal complexes. Considerable efforts have been undertaken to develop synthetic receptors for their selective molecular recognition. Ammonium ions are ubiquitous in chemistry and molecular biology.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
